
Industry committed to continue its investment in research and development to make animal-free toxicology a reality
SEURAT-1 is currently the largest European research initiative that aims to fundamentally change the way we assess the safety of chemicals. The goal is to move away from safety assessment relying on animal experiments towards a paradigm based on using mechanistic understanding of toxicology to intelligently integrate leading edge computational and in vitro tools into systems that support predictive safety assessment.
On 5 September 2013 in a dedicated workshop in Brussels, SEURAT-1 presented to stakeholders its latest scientific achievements delivered midway through its 5 years programme. The 50 participants, representing industry, policy makers, regulators and animal welfare groups appreciated the presentations that explained in a practical and accessible manner how new insights into mechanistic toxicology are being translated into solutions for safety assessment. Highlights included microfluidic bioreactors (so called “organ on a chip”) for long-term cultivation and analysis of human cells, toxicity assays based on human stem cells produced by reprogramming adult cells, and computational models for predicting safety thresholds.
The lively discussion that followed these presentations underlined:
- the strong interest of industry in this research work and its willingness to continue its investment in research and innovation enabling ultimately to deploy new safety assessment solutions in various industry sectors (pharma, cosmetics, food, …)
- the need to inform and involve the regulators on a much larger scale to make them aware of the changes on the horizon in the field of alternative animal-free human safety assessment methods
- the willingness of other private and public research initiatives in this field to cooperate and exchange knowledge, avoiding the duplication of effort and supporting faster progress
- the impact of the revolutionary concepts promoted by SEURAT-1 (and others), not only on safety assessment, but also for protection of human health in general, by prevention of e.g. diseases triggered by environmental factors
- the need for a global research strategy allowing to identify the next steps of the required long term research and innovation effort
 SEURAT-1 has developed such a global research strategy and published it in its Annual Reports, the volume 3 of which was presented during this stakeholder event. This report also provides a comprehensive overview of the research programmeand includes articles from international experts who provide their opinion on the state-of-play in regulatory safety assessment and the challenges and opportunities that lie ahead. The SEURAT-1 Annual Reports are publically available in the Library section. A hardcopy of Annual Report volume 3 can also be ordered free of charge on this website.
SEURAT-1 has developed such a global research strategy and published it in its Annual Reports, the volume 3 of which was presented during this stakeholder event. This report also provides a comprehensive overview of the research programmeand includes articles from international experts who provide their opinion on the state-of-play in regulatory safety assessment and the challenges and opportunities that lie ahead. The SEURAT-1 Annual Reports are publically available in the Library section. A hardcopy of Annual Report volume 3 can also be ordered free of charge on this website.
The European Partnership for Alternative Approaches to Animal Testing (EPAA), with a vision of replacement, reduction and refinement (3Rs) of animal use for meeting regulatory requirements through better and more predictive science, joined forces with SEURAT-1 to organize this stakeholder event thereby ensuring broad dissemination through its extensive member-network.
 |
 |
|
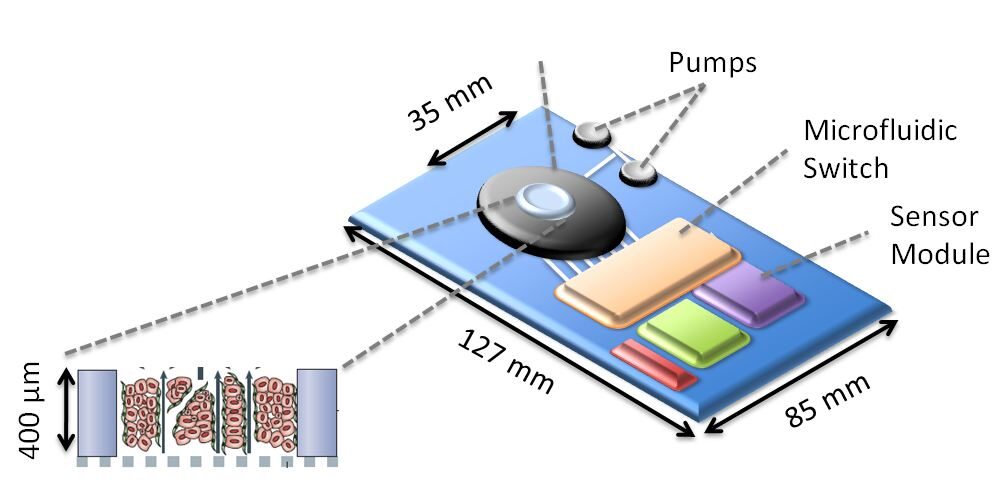
| Prototype of a microfluidic bioreactor used for “organ-on-a-chip” culture. A few square cm suffice to simulate a human liver which is then used to study a test compounds’ toxicity. Courtesy of Dr. Yaakov Nahmias, HeMiBio project within SEURAT-1. | This robot is able to produce billions of cells derived from reprogrammed human cells ; a capacity of producing such cells in large quantities is a prerequisite for animal free human safety assessment solutions, Source: I-stem (Inserm U861) |
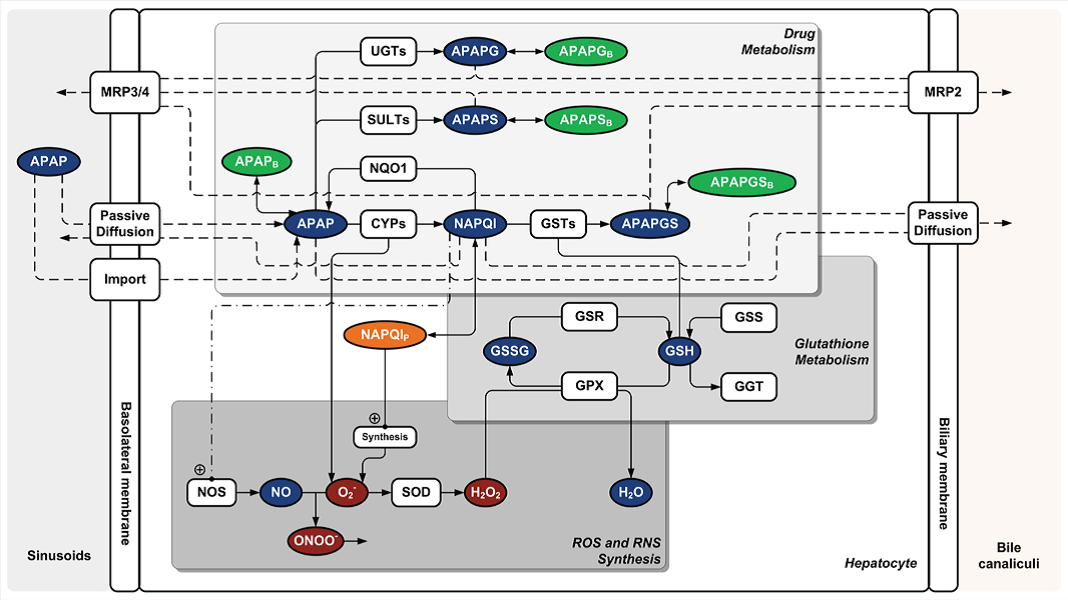
| Computer modelling is a key technology to enable the implementation animal free human safety assessment solutions |
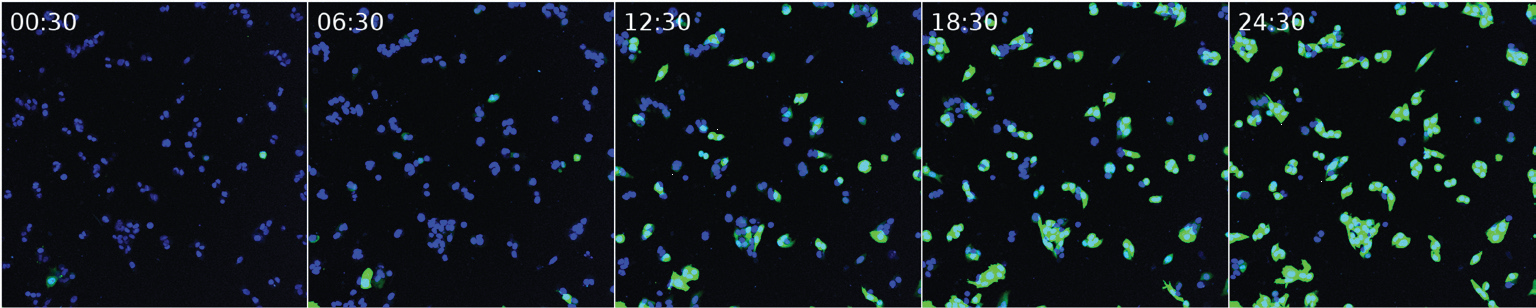
| Reporter cells: these cells used for human safety assessment have been engineered in such a way that they appear fluorescent green when they are exposed to a toxic substance |

Sara holds the esteemed position of Lead Publisher at SEURAT, overseeing the curation and dissemination of content centered on health and ethical consumer choices. With her rigorous editorial standards and an acute understanding of the health supplement industry, she ensures the delivery of accurate, insightful, and relevant information to the platform’s discerning audience. Her commitment to promoting cruelty-free practices has solidified the website’s reputation as a trusted source for professional and ethical insights in the sector.
